Omicron进荷兰?南非班机61确诊:几可确定有新变异株
11/27/2021

荷兰卫生当局今天表示,从南非飞抵阿姆斯特丹的2架航班中,61名COVID-19(2019冠状病毒疾病)筛检阳性的旅客中,据信至少「有一部分人」感染的是新的Omicron变异株。
路透报导,荷兰为预防Omicron入侵,26日禁止所有来自非洲南部的班机降落,但禁令宣布之前,荷兰皇家航空(KLM)2架分别从南非开普敦与约翰尼斯堡起飞的客机已抵达阿姆斯特丹史基普机场(Schiphol Airport),这两架班机上共载有624人。
荷兰国家公共卫生及环境研究院(National Institute for Public Health, RIVM)稍后发布声明表示,「完成COVID-19筛检、结果呈阳性反应的人之中,一部分可能已经染Omicron。」
发言人说,他们「几乎肯定」这些病例为Omicron,但需要进一步分析。RIVM表示,结果将在明天公布。
荷兰卫生局(Dutch Health Authority, GGD)则发布声明说:「我们目前知道的是,61人的筛检结果呈阳性反应,531人为阴性反应。」
卫生局表示,筛检结果呈阳性反应的人,有症状者一律在旅馆隔离7天,无症状者隔离5天;筛检呈阴性反应的人,入境者必须居家隔离,过境者则可继续走接下来的行程。
●登机都有阴性证明 KLM:无法解释
荷兰皇家航空的发言人说,乘客在开普敦与约翰尼斯堡上机前,都有出示筛检阴性或疫苗接种证明。
如今有这么多人确诊,发言人说,「我们无法做出解释」,有可能是接种疫苗者有人确诊,也可能当中「从阴转阳」的人数出奇的多。
法新社报导,乘客表示,为了采检,他们在入境大厅苦等好几个小时。他们也通报说,现场许多人没有带口罩,也没有维持社交距离。
●隔离筛检折腾一晚 乘客:苦等煎熬
根据乘客齐默曼(Paula Zimmerman)记录了她这一个晚上的经历,并将影片上传推特(Twitter)。她表示,苦等数小时之后,她终于收到筛检阴性的通知,但接下来仍得待在家中隔离5天。
齐默曼告诉法新社,机上的人「从停机坪被带到隔离的房间,等了4个小时才开始进行筛检」。
她说,「机上每个人都在那里」,且几乎都没有遵守社交距离规范。
她发布的最后一支影片显示,还有一排乘客等着筛检,另外还有一些人已经睡倒在长椅或地板上。
她推文说:「当局的组织能力很糟糕,这很令人遗憾,希望已汲取到相关经验供未来参考。」
防Omicron变异株 澳门禁21天内到过非洲8国人员入境
世界新闻网
11/27/2021

为防范新冠变异株Omicron入侵,澳门政府今晚宣布,从28日零时起,禁止民航机搭载任何在21天内曾到过南非、博茨瓦纳等8个非洲国家的人士前来澳门。
据澳门新型冠状病毒感染应变协调中心宣布,这8个非洲国家是南非、博茨瓦纳、津巴布韦、纳米比亚、莱索托、史瓦帝尼、莫桑比克与马拉威。
南非新变异株Omicron为防范疫情投下变量,香港出现首例亚洲2例确诊个案。港府26日深夜宣布全球最严的防疫措施,27日零时起,禁止21天内曾在南非、博茨瓦纳、纳米比亚、莱索托、史瓦帝尼、津巴布韦、马拉威、莫桑比克等非洲南部8国逗留的非香港居民入境。
香港通报非洲变种病毒境外输入案例
11/26/2021

香港通报两名境外输入冠病病例,他们所携带的病毒基因排序与早前在南非发现的B.1.1.529冠病新变种相似。
香港政府新闻网昨(25日)晚公布,香港大学的全基因组测序分析结果显示,有两名病患的基因排序十分相似,并带有新出现的病毒株谱系B.1.1.529,与在南非和博茨瓦纳发现的排序相似。卫生署卫生防护中心说,其中一人从南非抵港,属于源头个案。
《明报》早前报道,这名从南非抵港病患是一名36岁印度籍男子,本月11日抵达香港后入住富豪机场酒店的5112号房间。他在检疫第三天(13日)的检测结果呈阳性,病毒量高。

另一名病患则是62岁中国籍男子,10日由加拿大抵港后同样入住富豪机场酒店同层斜对面的5111号房间,检疫第九天(18日)的检测结果呈阳性。
防护中心已安排本月11至14日入住该酒店5111和5112号房间左右相连三个房间(即5105至5120号共12个房间)的12人,到检疫中心接受14天强制检疫,暂时未有相关确诊个案。
港府专家顾问袁国勇早前对上述病例进行调查后发现,先确诊的印度籍男子戴气阀口罩,喷出病毒没有被过滤,开酒店房间门取餐或倒垃圾时,有一半的时间没戴口罩,且房外空气流动不足,导致病毒停留,预测带病毒的空气流出走廊,导致斜对面房男子感染。袁国勇奉劝市民尤其外国返港者,改用外科口罩。
南非新变种病毒撼动全球市场 它有何不同、接下来要注意什么
文 / 张佳莹
11/26/2021
(早报讯)南非发现一种名为B.1.1.529的新变种冠病病毒,令当地官员忧心忡忡。新的变种病毒可能助长许多国家的疫情,增加卫生系统的负荷,甚至使重新开放经济和边境的努力变得更加复杂,因此造成全球市场周五出现一波避险浪潮。
据彭博社报道,初步对新变种冠病病毒的了解如下:
1. 这个变种病毒有何不同?
科学家们说,B.1.1.529的刺突蛋白(spike protein)带有大量变异,这在病毒进入体内细胞中起着关键作用。研究人员仍在试图确定它是否比先前的变种病毒更容易传播或更致命。
2.它从哪里来?
目前只能猜测。英国伦敦大学学院(UCL)遗传学研究所的一名科学家说,可能是在一名免疫功能低下的人体中,于慢性感染期间演变而来。此人可能是未经治疗的人体免疫缺陷病毒(HIV,俗称爱之病病毒)患者。南非有820万人感染HIV,为全球之最。贝塔(Beta)变种病毒是去年在南非发现的一种变种,也可能来自HIV感染者。
3.它的传播有多广?
截至周四,南非已发现近100起病例,它在当地已成为新增病例中的主要毒株。但《金融时报》援引当地卫生官员的话说,早期聚合酶链反应(PCR)检测结果显示,周三在包括约翰内斯堡在内的省分报告新增的1100起病例中,90%是由新变种病毒引起的。邻国博茨瓦纳的官员周一报告,有四起病例是完成接种疫苗的人。在香港,一名来自南非的旅客被发现带有这个变种病毒,另一起病例则是正在接受隔离的人。
4.外界的反应如何?
有关新变种病毒的消息震动了周五的市场,亚洲与旅游相关的股票跌幅最大,因为投资者预计它将对旅游产生负面影响。英国暂时停止来自非洲六个国家的航班入境;澳大利亚表示,如果情况升级,不排除收紧来自非洲南部旅客的入境规定。印度加强了对来自南非、博茨瓦纳和香港入境旅客的筛查。通常被视为避险资产的日元兑美元上涨0.4%,南非兰特兑美元汇率跌至一年低点。
5.这有多令人担忧?
目前还言之过早。世界卫生组织表示,新变种病毒可用的完整基因组序列不到100个,可能会增加研究所需的时间。病毒一直在变异,这些变化有时会使病毒变弱,有时会使其更擅长逃过抗体。
6.接下来要注意什么?
世界卫生组织决定周五(11月26日)召开会议讨论B.1.1.529,并决定是否将其正式列为“需留意变异株”抑或是“需关注变异株”。如果是如此,将根据世界卫生组织命名计划以希腊字母为这个变种病毒命名,可能是“nu”。各国政府也可能会针对该变种病毒最新情况对边境和旅行管制采取行动。
捷克总统冠病检测结果呈阳性 此前已接种三剂冠病疫苗
文 / 张佳莹
11/25/2021

(早报讯)捷克总统办公室周四(25日)晚发布声明说,总统泽曼当天冠病检测结果呈阳性。
声明说,泽曼上个月因病入住布拉格的中央军事医院。25日早上出院后回到布拉格西郊的拉尼城堡,当天下午接受冠病检测结果呈阳性,之后再次入院。在接受冠病治疗期间,泽曼将暂停官方活动。
据当地媒体报道,泽曼目前尚未出现相关冠病症状,他此前已接种三剂冠病疫苗。
捷克卫生部25日发布的数据显示,截至24日,该国累计冠病确诊病例超过200万起。
欧洲疫情一幕:这国现尸体堆积如山惨况
文章来源: 看世界杂志
11/23/2021

11 月 22 日,罗马尼亚的布加勒斯特大学医院里出现了这样一幕:
可容纳 15 具尸体的停尸房已经全部被塞满,门外还有堆积如山的尸体正在排队等候。这些尸体被裹在黑色塑料袋里,躺在一排排医院轮床上,挤满了整个医院走廊。
罗马尼亚布加勒斯特大学急救医院,一名医务人员在拥挤的新冠隔离室中行走
布加勒斯特大学医院是罗马尼亚首都最大的治疗新冠的医疗机构,但在新一轮的疫情冲击下,医疗系统再度濒临崩溃,医护人员也告诉 CNN 记者,他们从没想过会再发生 ” 将整个家庭送入坟墓的灾难 “。
进入 11 月后,欧洲日均录得的新冠确诊以及死亡病例数,超过了同期全球新增确诊及死亡病例数的一半。曾因处理疫情得当而受到西方媒体肯定的德国,11 月 17 日的单日新增确诊病例达到 68366 例,创下德国暴发疫情以来的最高纪录。邻国荷兰也从 11 月 16 日开始,每日新增病例都超过了 2 万,并且连续第三天创下新纪录。而在受到第五波疫情冲击的法国,11 月 17 日当天的新病例数也超过了 2 万,至今都没有缓和的迹象。

布加勒斯特街边的广告牌上印着医务人员的抗疫身影,下方的标语写着:” 他们令人窒息。他们在乞求我们。他们在后悔。”
11 月 4 日,世界卫生组织宣布,欧洲再次成为新冠病毒的重灾区。这意味着欧洲进入第四波疫情。世卫组织欧洲区域办事处主任汉斯 · 克鲁格在接受媒体采访时表示,根据目前欧洲的新冠疫情发展态势,若不采取紧急防疫举措,到明年 3 月,欧洲或将有 50 万人死于新冠肺炎。
欧洲今年的疫情,似乎进入一个跟去年一样的循环:年初天气寒冷,确诊病例数节节攀升,进入夏天后疫情稍缓,各种隔离措施相继解除,之后进入年底,疫情重新反扑。反复的解封和封城,似乎成为了疫情暴发以来各国开始习惯的日常,仿佛走钢丝一样,战战兢兢地走了两年。
英国《金融时报》把欧洲第四波疫情归咎于:天气转冷让人们的室内活动增加,以及欧盟部分国家的民众疫苗接受程度低。另外,尽管欧洲多国确诊人数比去年第二波要高,但是死亡率和入院数字并无激增。
欧洲疫情地图
面对第四波疫情,欧洲各国政府的应对措施与之前的最大差别,是打了疫苗的国民和没打疫苗的国民受到不同待遇。以奥地利为代表,11 月 17 日凌晨起,未打疫苗的奥地利国民将被限制出门,外出活动被局限于看病,或者采购生活必需品。
奥地利政府规定,这一限行措施为期 10 天,但不排除在疫情没好转前延续这一措施。
在第四波疫情中,比奥地利更早采取部分封城措施的欧洲国家,是荷兰。11 月 12 日,荷兰首相马克 · 吕特宣布了为期三周的 ” 娱乐场所关闭、非必需品商铺和服务业门店晚 6 时关门、生活必需品商铺晚 8 时关门 ” 的措施。

不过,相比去年两波疫情期间的措施,荷兰今次封城显得更 ” 软 “:学校、戏院和电影院都没关闭,一些静态的公开活动(譬如讲座和论坛)也继续举行,但是展览和商业展会等造成人员在密闭空间流动的活动被叫停。
荷兰的新冠确诊人数在 10 月上旬就开始急剧攀升,奥地利稍晚暴发,连拉几个高线也让人焦急。
按照世界卫生组织在欧洲地区办公室的说法,荷兰和奥地利分属两个不同的疫苗注射梯队:
奥地利、捷克、斯洛伐克和波兰等中欧国家,属于疫苗注射不理想的国家;排在其后的罗马尼亚(34.5%)和保加利亚(23.0%),是整个欧盟当中疫苗注射率最低的两个国家。

相比之下,德、法、比、荷等西欧多国的疫苗注射率,均达到欧盟平均水平以上。
然而,这些国家的每日新增病例曲线自 10 月起同样往上攀升。葡萄牙和西班牙,以及马耳他和意大利这几个南欧国家,算是攀升曲线幅度相对平缓的国家,其中葡萄牙的疫苗注射率达到 87.6%,是全欧洲第一。
纵观整个欧洲的数据,一个让人沮丧的现象是:疫苗注射率低的国家病例猛增,带动疫苗注射率高的国家病例攀升,整个欧洲疫情严重程度从西到东依次递增。
盯紧未打疫苗者
面对重新暴涨的新冠疫情,欧洲多国政府把矛头对准未注射疫苗的国民。
随着疫情折腾近两年,欧洲各国的封城措施也与过去疫情刚暴发时的 ” 一刀切 ” 显得不同。
时至今日,封城政策制定的一个重要准绳,便是疫苗在本国的注射率。
奥地利防疫政策背后,自然是有 ” 用限行措施倒逼国民打疫苗 ” 的企图。据统计,这项措施将会让大概 200 万名奥地利公民出门受限制,而该国已注射两针疫苗的国民比例为 63.6%,略低于欧盟整体标准。
德国联邦政府也开始放出风声,采取类似奥地利的专门针对没注射疫苗国民的限行措施。
柏林地方政府在 11 月 17 日宣布,进入酒吧和餐馆等地方,必须持有已注射疫苗的证明;德国卫生部长杨斯 · 斯波恩也表态,可能会跟随柏林地方政府采取相似措施。

除此之外,希腊、拉脱维亚、爱沙尼亚等国,都相继出台了针对未注射疫苗国民的限行措施。然而,在一些疫苗注射率高于欧盟平均水平的国家,疫情依然不容乐观。譬如法国有着 68.7% 的疫苗注射率,但是自 11 月 11 日起,法国日增确诊人数又再次过万,甚至达到两万。
即便是在执行健康通行证最严厉和注射疫苗率排在欧洲第五的意大利,新增病例依然在一些意想不到的城市大规模出现。
在意大利港口城市的里雅斯特(Trieste),经过一场反对强推健康通行证的示威后,该城市出现了新冠疫情在周末急剧扩散的现象。的里雅斯特当地的医院向媒体反映,该市最近收治的新冠病人,有 90% 都没有注射疫苗,并且都跟反疫苗游行有关联。
10 月 16 日,罗马的抗议者们手持棍棒和金属棒,冲进意大利劳工总联合会的总部。他们砸毁了办公室,抗议政府要求员工接种新冠疫苗或接受阴性检测的规定 。
欧洲当地一些学者批评,政府的第三剂 ” 加强针 ” 疫苗推广政策不力,导致了日增病例不断刷新纪录。他们把以色列视作可以效仿的 ” 榜样 “:早在 7 月份,以色列便开始给超过 60 岁的公民注射第三剂 ” 加强针 ” 辉瑞疫苗——而自 10 月 18 日起,以色列的日增新冠病例数开始跌破 1000,曲线不断往下走。
借鉴 ” 加强针 ” 成功经验
在 9 月份联合国大会上,以色列总理贝内特发表演说,宣布以色列 ” 率先开打加强针 “。以色列所谓的 ” 加强针 “,是基于以色列卫生部在夏天的一项研究结果得出的举措。
该研究发现,人体对新冠病毒的抗体水平,在注射了两针辉瑞疫苗 6 个月后将显著下降;而年龄在 60 岁以上的长者,在注射了第三针疫苗后,其体内抗体水平将在 10 天内提高 6 倍,并且能有效防止新冠病毒引起的严重症状。
如果从近半年的日增确诊数据看,以色列的感染曲线的确在 10 月持续往下掉,进入 11 月后日均确诊病例不超过 300。美国威斯康星大学病理学教授大卫 · 欧康奈认为,” 加强针 ” 应该成为各国防疫的标准措施。

然而,《自然》期刊同时也指出,以色列的疫苗注射计划具有独特性:以色列国土狭小,并且在独特的地缘政治格局中拥有丰富的全民应急设施,全国公共医疗系统并不庞大,但是能深入社区基层。
而且,” 加强针 ” 同时也面临一个伦理问题:在疫苗数量有限的现实中,疫苗是应该分配给从没得到一针疫苗的公民,还是已经注射了两针的公民?以色列作为一个可供借鉴的 ” 活的实验室 “,并不完全跟世界其他国家的需求吻合。
对于欧洲国家来说,以色列先例的一个可借鉴之处,是打完了 ” 加强针 ” 后,不但扭转了第四波的曲线,更避免了要施加封城政策带来的痛苦。到目前为止,以色列政府在国内已经取消了年初采取的各种限制商业活动的行为,但前提是执行健康通行证政策,以及公众场所必须戴口罩的措施。
在疫苗注射率相对较高的法国,总统马克龙宣布,年满 65 岁及以上的长者必须注射第三针疫苗,才能获得出行所需的健康通行证。与此同时,挪威和意大利也将在 12 月推出高龄长者的 ” 加强针 ” 计划。

以色列本 · 古里安大学教授纳达夫 · 大卫朵维奇认为,欧洲目前的第四波疫情很像以色列在 7 月经历的第四波,德尔塔变种加上两针疫苗半年后出现的抗体衰减,是如今第四波在 2021 年冬天卷席欧洲的原因。而欧洲国家要避免去年那样执行严酷的封城措施,就不得不跟随以色列的步骤,不断推广 ” 加强针 ” 的注射。
在现行技术还没出现更多突破之前,也许每隔一段时间推广一次 ” 加强针 “,就是人类跟新冠病毒较量的最好手段了。
澳洲将采购30万份默克实验性抗冠病口服药疗程
文 / 陈慧璋
10/05/2021
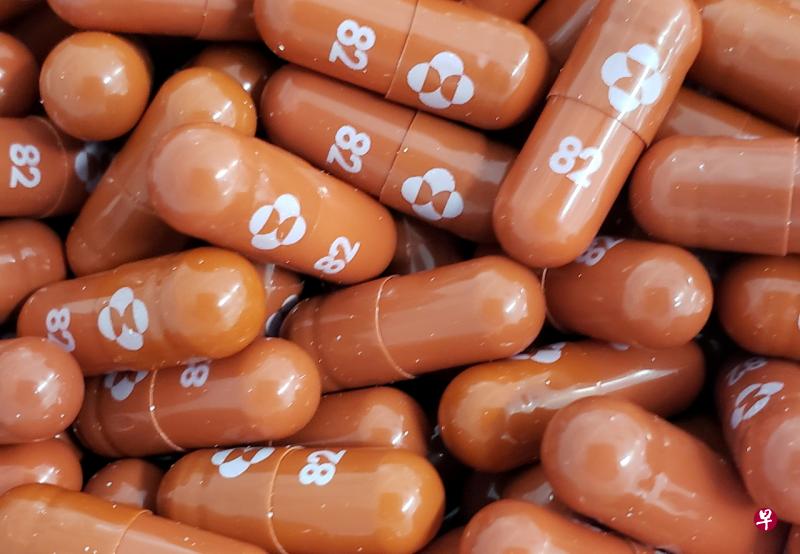
(早报讯)澳大利亚总理莫里森周二(5日)宣布,澳洲将向美国药厂默克公司(Merck & Co)购买30万份实验性抗冠病口服药疗程。
路透社报道,这款名为Molnupiravir的药物是第一款抗冠病口服药。专家表示,如果Molnupiravir获药物监管当局通过,将有望使冠病死亡病例和住院病例减半。
莫里森周二接受澳洲九号新闻网访问时说,10粒Molnupiravir为一个疗程。每名成年冠病患者必须连续五天服用Molnupiravir,每次服两粒。
莫里森说:“Molnupiravir将允许我们与冠病病毒共存。”
他还表示,一旦获得澳洲药品监管局批准,澳洲应可在明年年初收到30万份Molnupiravir。
默克公司预计到今年年底可生产1000万份Molnupiravir疗程。
Molnupiravir的临床试验是在菲律宾进行,韩国、泰国、台湾和马来西亚均表示,正与默克公司磋商采购这款口服药的事宜。

北美法律公益讲座安排
时间:周二到周五 晚间
5:30-7:00(西部)
8:30-9:30(东部)
第二天西部时间早上9:00重播
周二: 如何准备遗嘱文件(遗嘱workshop)
周三: 数据泄露和个人身份保护&事业机会说明会
周四: 移民和留学生常见法律问题
周五:小企业企业常见法律问题&事业机会说明会
Zoom 6045004698,
密码:进群获取

默沙东抗疫口服药可降低50%住院或死亡风险
文 / 麦可欣
10/02/2021
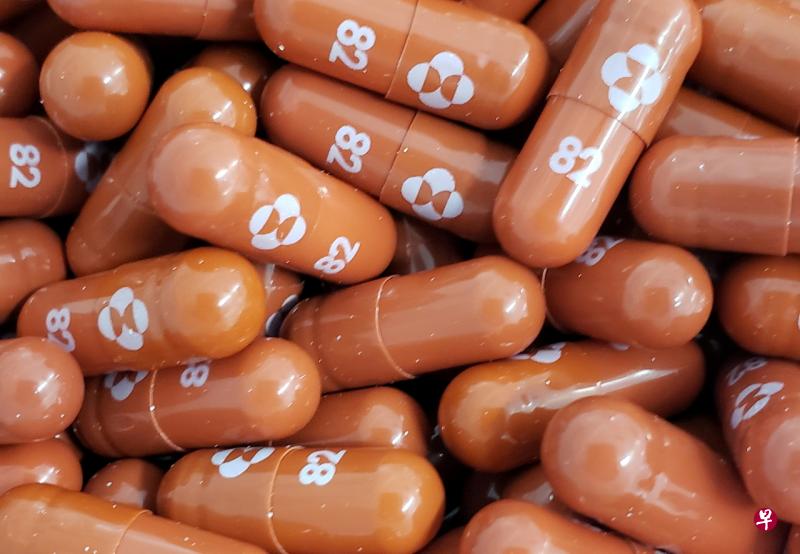
(早报讯)美国默沙东药厂(Merck & Co Inc)周五(10月1日)公布,研发中的冠病实验口服药“molnupiravir”,可使重症高风险患者的住院或死亡率降低约50%。默沙东表示,将向美国当局申请紧急授权使用,一旦获批准,将是全球首款抗冠病口服药。
默沙东药厂发表声明说,:“根据期中分析,服用molnupiravir的染疫患者中,至29天有7.3%住院;而服用安慰剂的患者,至29天有14.1%住院或死亡。”
默沙东药厂计划针对这个口服药尽快向美国提出紧急使用授权,并且向全球监管机关递交申请。由于临床实验结果证实有疗效,在外部监测人员建议下,第三期临床试验提早喊停。
Merck says its new Covid pill reduces the risk of hospitalization, death by half for some patients
By Chloe Taylor
10/02/2021
KEY POINTS
- A phase 3 trial of Merck and Ridgeback Biotherapeutics’ oral antiviral treatment molnupiravir showed it reduced the risk of hospitalization or death by around 50% in Covid patients.
- Merck plans to seek emergency use authorization in the U.S. and submit marketing applications to other global drug regulators.
- If authorized by regulatory bodies, molnupiravir could be the first oral antiviral medicine for Covid.
- “The company, when they briefed us last night, had mentioned that they will be submitting their data to the FDA imminently,” White House chief medical advisor Dr. Anthony Fauci said at a Covid briefing Friday.
Merck and Ridgeback Biotherapeutics said Friday they’ve developed a drug that reduces the risk of hospitalization or death by around 50% for patients with mild or moderate cases of Covid.
The companies plan to seek emergency authorization for the antiviral Covid treatment after the medicine showed “compelling results” in clinical trials.
The drug, molnupiravir, is administered orally and works by inhibiting the replication of the coronavirus inside the body.
An interim analysis of a phase 3 study found that 7.3% of patients treated with molnupiravir were hospitalized within 29 days. Of the patients who received a placebo, 14.1% were hospitalized or died by day 29. No deaths were reported in patients who were given molnupiravir within the 29-day period, while eight deaths were reported in placebo-treated patients.
“The news of the efficacy of this particular antiviral is obviously very good news,” White House chief medical advisor Dr. Anthony Fauci said at a Covid briefing Friday. “The company, when they briefed us last night, had mentioned that they will be submitting their data to the FDA imminently.”
“The FDA will look at the data and in their usual, very efficient and effective way, will examine the data as quickly as they possibly can, and then it will be taken from there,” Fauci said.
All 775 trial participants had laboratory-confirmed symptomatic Covid-19 and were randomly given molnupiravir or a placebo within five days of symptoms.
Every participant was unvaccinated and had at least one underlying factor that put them at greater risk of developing a more severe case of the virus. The most common risk factors included obesity, being over age 60 and having diabetes or heart disease.
The phase 3 part of the trial was conducted at more than 170 sites, in countries including the U.S., Brazil, Italy, Japan, South Africa, Taiwan and Guatemala.
Molnupiravir’s efficacy was not affected by the timing of symptom onset or patients’ underlying risk factors, the study showed. It also proved to be consistently effective in treating all variants of Covid, including the widely dominant and highly transmissible delta strain.
Adverse events were comparable in the molnupiravir and placebo groups, with around 10% reporting adverse events. Just 1.3% of the molnupiravir group discontinued therapy due to an adverse event — less than the 3.4% of the placebo group who did so.
Recruitment into the study is being stopped early due to the positive results, at the recommendation of an independent Data Monitoring Committee and in consultation with the U.S. Food and Drug Administration.
Merck is also testing molnupiravir in a separate global phase 3 study to evaluate its efficacy in preventing the spread of Covid within households.
‘Profound impact’
Robert M. Davis, CEO and president of Merck, said in a press release Friday that the company would do everything it can to bring molnupiravir to patients as quickly as possible.
“With these compelling results, we are optimistic that molnupiravir can become an important medicine as part of the global efforts to fight the pandemic,” he said.
Ridgeback Biotherapeutics CEO Wendy Holman added: “With the virus continuing to circulate widely, and because therapeutic options currently available are infused or require access to a healthcare facility, antiviral treatments that can be taken at home to keep people with Covid-19 out of the hospital are critically needed.”
“We are very encouraged by the results from the interim analysis and hope molnupiravir, if authorized for use, can make a profound impact in controlling the pandemic,” she said.
Emergency use authorization
Merck said Friday it plans to seek emergency use authorization for the drug in the U.S. as soon as possible. The company also plans to submit marketing applications to other international drug regulators.
If authorized by regulatory bodies, molnupiravir could be the first oral antiviral medicine for Covid. Antiviral treatments now in use, such as remdesivir, are administered intravenously.
Merck has already begun producing molnupiravir. The pharmaceutical giant expects to produce 10 million courses of treatment by the end of 2021, and more doses in 2022.
The company agreed earlier this year to supply the U.S. with around 1.7 million courses of molnupiravir if it receives emergency use authorization or full approval from the FDA. The federal government also has the option to purchase additional doses if the drug is approved, White House coronavirus response coordinator Jeff Zients said at Friday’s briefing.
Merck has also entered supply and purchase agreements for the drug with other governments — pending regulatory authorization — and is in discussions with other governments about the supply of molnupiravir.
The company said it plans to implement a tiered pricing approach based on World Bank country income criteria to ensure molnupiravir can be accessed globally. Merck previously announced that it had entered into nonexclusive voluntary licensing agreements for molnupiravir with generic manufacturers, a move intended to assist low and middle-income countries in gaining access to the treatment. Those agreements are also pending approvals or emergency authorization by local regulators.
Profit share
Ridgeback received an upfront payment from Merck as part of the companies’ development of molnupiravir. The company is also eligible to receive contingent payments depending on developmental and regulatory approval milestones.
Profits arising from the collaboration will be split between Merck and Ridgeback equally.
Supreme Court Justice Brett Kavanaugh tests positive for COVID-19
Kavanaugh shows no symptoms, is fully vaccinated, court says
By Houston Keene | Fox News
10/01/2021
Supreme Court Justice Brett Kavanaugh tested positive for COVID-19, the court announced Friday, noting that he’s fully vaccinated and showed no symptoms.
Kavanaugh learned of the positive test Thursday evening, ahead of Justice Amy Coney Barrett’s ceremonial investiture Friday morning, according to a news release from the court.

The release added that Kavanaugh’s wife and daughters, all fully vaccinated, tested negative.
“On Thursday, per the Court’s regular testing protocols, Justice Kavanaugh had a routine Covid test ahead of Justice Barrett’s investiture on Friday,” the release said. “On Thursday evening, Justice Kavanaugh was informed that he had tested positive for Covid-19.”
“He has no symptoms and has been fully vaccinated since January. Per current Court testing protocols, all of the Justices were tested Monday morning prior to conference, and all tested negative, including Justice Kavanaugh,” it continued.
Barrett and Kavanaugh will be sitting at opposite ends of the table when oral arguments resume in the Supreme Court on Monday.
Kavanaugh ran in the Capital Challenge Road Race on Wednesday, with his team winning the three-mile race’s judicial division with a 25 minute time.
Several lawmakers, judges and members of the media also participated in the race. Fox News’ Sandra Smith placed first in the Electronic Journalist – Female category while Fox News took first in the Electronic Media category.
COVID-19 breakthrough cases have hit Capitol Hill recently. Fully vaccinated South Carolina Republicans Sen. Lindsey Graham and Rep. Ralph Norman tested positive for the virus in August.
As of Friday, the U.S. has 3.7 million COVID-19 cases and almost 52,000 deaths reported within the last 28 days, according to Johns Hopkins University’s COVID-19 tracker.
1 in 500 US residents has died of Covid-19
By CNN Wire
9/15/2021
(CNN) — The United States has reached another grim milestone in its fight against the devastating Covid-19 pandemic: 1 in 500 Americans have died from coronavirus since the nation’s first reported infection.
As of Tuesday night, 663,913 people in the US have died of Covid-19, according to Johns Hopkins University data. According to the US Census Bureau, the US population as of April 2020 was 331.4 million.
It’s a sobering toll that comes as hospitals in the US are struggling to keep up with the volume of patients and more children are grappling with the virus. In hopes of managing the spread and preventing more unnecessary deaths, officials are implementing mandates for vaccinations in workplaces and masking in schools.
They are fighting against a sharp upward trend in cases and deaths: The US is reporting a more than 30% increase in average daily cases and a near tripling of average daily deaths over the past month, according to data from the US Centers for Disease Control and Prevention.
But with only 54% of the population fully vaccinated, the rate of people initiating vaccinations each day has declined over the past month.
Health experts have hailed vaccinations as the best source of protection against the virus, noting that the majority of people hospitalized with and killed by Covid-19 are unvaccinated. In Pennsylvania, from January 1 to September 7, 97% of the state’s Covid-19 deaths were among unvaccinated people, Pennsylvania’s acting secretary of health said Tuesday.
Another layer of strong protection, experts say, is masking.
The CDC recommends people — even those fully vaccinated — wear masks indoors in areas with substantial or high community transmission. More than 99% of the population lives in a county with one of those designations.
In Ohio, where children’s hospitals are overwhelmed with Covid-19 and respiratory cases, Gov. Mike DeWine is encouraging schools to issue mask mandates since the state legislature has told him it would overturn any mandate he issued.
“Reasonable people may disagree about a lot, but we can all agree that we must keep our children in the classroom so they don’t fall behind and so their parents can go to work and not take time off to watch their kids at home,” DeWine said.
The combination of masks and vaccinations is the way to keep children in school, Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, told CNN Tuesday.
“If you surround the kids with vaccinated people and you have everybody wear a mask, you can get a situation where the children will be relatively safe in school,” Fauci told CNN’s Jake Tapper.
Fight brewing over vaccine mandates
In the effort to manage the spread of the virus, many officials and experts have promoted vaccine mandates — but others are opposing such measures.
New York issued an order in August requiring all health care workers be vaccinated against Covid-19 by September 27. But on Monday, 17 Catholic and Baptist medical professionals filed a federal complaint seeking to prevent the state from enforcing the mandate, saying they oppose getting the vaccine for religious reasons.
On Tuesday, a federal judge issued a restraining order temporarily suspending New York state from enforcing its vaccine mandate if health care workers claim a religious exemption.
Because the mandate does not require health care workers to receive their first dose of the vaccine until September 27, the judge’s order states the temporary restraining order “does not, as a practical matter, go into effect until that date.”
A hearing is scheduled for September 28.
After the ruling, New York Gov. Kathy Hochul’s press secretary, Hazel Crampton-Hays said in a statement that the governor is considering all legal options.
“Governor Hochul is doing everything in her power to protect New Yorkers and combat the Delta variant by increasing vaccine rates across the State,” Crampton-Hays said.
In Los Angeles, despite a mandate that all city employees be inoculated against the virus, nearly a quarter of the police force is seeking an exemption, according to Mayor Eric Garcetti’s office. Those who are not vaccinated will be required to show evidence of weekly testing and a negative COVID result if regularly reporting to work.
By November 1, Nevada workers who serve “vulnerable populations” must show proof of vaccination under a new emergency regulation passed Tuesday.
New hires must have at least one dose by their start date and must follow through on the required vaccination schedule to remain employed. Workers are allowed to ask for a medical or religious exemption.
Booster meeting won’t be a slam dunk
On Friday, the US Food and Drug Administration (FDA) will meet to discuss whether most Americans need a booster of their Covid-19 vaccine.
Unlike other meetings to discuss the vaccine, this one, with requests from Pfizer to authorize a third dose for most people, won’t be a slam dunk.
“This will be much messier than in December,” said Dr. William Schaffner, an infectious disease specialist at Vanderbilt University. The FDA committee was quick to recommend authorization of vaccines made by Pfizer and rival Moderna last December.
When the FDA’s Vaccines and Related Biological Products Advisory Committee meets Friday, it will be presented with dueling data, some of it suggesting there’s a need for boosters, but other pieces of data suggesting there is no such need.
Three separate articles published last week in the CDC’s Morbidity and Mortality Weekly Report suggest that we don’t need boosters.
On the other hand, an Israeli study found that over time, the vaccines’ power to keep people from getting very sick with Covid-19 diminished. Looking at illnesses in the second half of July, that study found that those who’d received their second dose of Pfizer’s vaccine in March were 70% more protected against severe disease than those who received the second shot in January.
President Joe Biden announced plans last month to begin administering booster doses next week. While she wouldn’t say directly if that date would be met, CDC Director Rochelle Walensky said Tuesday she is hopeful about the timeline to get doses administered.
If the booster does get approved, experts will still have to wait and see how much protection is added by the third dose.
“I would hope that that would sustain us for an extended period of time, but I don’t know that right now,” Fauci said. “We’re just going to have to do the boost, and then follow people long enough to determine what the durability of that protection is.”




